Editor’s note: Yilian Yuan is director, marketing analytics, IMS Management Consulting, Fairfield, Conn.
Few industries spend as much on research and development as the pharmaceutical industry. Said another way, few have as much at risk when bringing a new product to market. The top 10 pharmaceutical companies in the world each spend an average of $3.8 billion a year on R&D, and the cost of miscalculating what the return will be on that investment could be disastrous. Thus, the pharmaceutical industry has developed very sophisticated methods of forecasting the market potential for a new product. Forecasting is an integral part of pharmaceutical business planning and provides critical information so that informed decisions can be made on how to allocate R&D and marketing resources.
Depending on the situation, there are different ways to forecast new product market potential. One is analog and event-based, another is patient-based, and sometimes the two approaches are combined. This article serves as a primer on the analog-based approach to forecasting for new pharmaceutical products in development and outlines the major steps involved.
Market overview and situation analysis
The first step in performing any forecast is to thoroughly understand the therapeutic market in question and its competitive dynamics. This entails analyzing:
- the current competitors in terms of their prescription volume and product share trends, product characteristics, labeling information, supplemental studies and additional indications;
- the promotional mix, share of voice and pricing trends for each of the competitors in the market;
- epidemiological trends over time and any changes in treatment guidelines.
For example, to forecast product market potential in the cholesterol treatment market, it is necessary to understand current treatment guidelines and the size of the potential patient population. In 2002, according to the National Health and Nutrition Examination Survey (NHANES; 1999-2002), over 100 million Americans had total cholesterol levels of 200 mg/dL or higher (borderline high-risk). The latest guideline published by the U.S. government’s National Cholesterol Education Program (www.nhlbi.nih.gov/chd ) calls for even more aggressive drug treatment and a lower LDL target value.
Several drug classes are being used to manage lipid problems: bile acid sequestrants, fibric acid derivatives, cholesterol absorption inhibitors, and statins. Statins and statins combined with cholesterol absorption inhibitors are the most popular drugs to lower cholesterol levels, such that about 150 million prescriptions were dispensed in 2004 for them, according to IMS Health’s National Prescription Audit. Within this subgroup of drugs, Merck’s Mevacor, which was introduced in 1987, was the first to enter the market. Mevacor lost patent protection in 2001 and now has been replaced by generics. The current leading product is Lipitor, which was launched in January of 1997 by Pfizer, and now has about 50 percent of the market share of this subgroup. Other major products are Pravachol, launched in November 1991 by Bristol-Myers Squibb; Zocor, launched in January 1992 by Merck; Crestor, launched in August 2003 by AstraZeneca; and Zetia, launched in November 2002 and co-promoted by Merck and Schering-Plough (Figure 1).

Analog selection and analysis
Analogs play an important role in forecasting a new product’s market potential since there is obviously no performance history that can be referenced for the product of interest. The best analogs are those that are similar to the new product in terms of disease type, order of entry into the market, advantages and disadvantages over existing treatment choices (such as efficacy, dosing, mechanism of action, side effects, safety, etc.), price relative to existing drugs, and level of promotional support. However, it is almost impossible to find analogs that match the product of interest in all of these respects. The solution is to combine several analogs in order to create a baseline forecast for a new product.
Baseline forecast
The baseline forecast is developed in two stages. The first stage involves estimating the growth in prescription volume within the therapeutic based on historic levels and assuming that the future will be like the past. The second stage entails analyzing the performance of the analogs and developing a forecast for the market share of the new product accordingly. Because it is very hard to find analogs that precisely match the product of interest in terms of market environment or product attributes and differentiation, it is often necessary to modify the share curves of the analogs in order to fit the situation at hand.
Statistical models, such as time series ARIMA (autoregressive integrated moving average) models, regression models and exponential smoothing, are often used to develop the baseline forecasts for the market volume. For detailed descriptions of statistic techniques and their applications in forecasting the market volume, please see the book Forecasting: Methods and Applications (Spyros Makridakis, Steven Wheelwright, and Rob Hyndman, 1998).
For the purpose of establishing a baseline forecast for a new product, an analog’s absolute prescription volume is not useful since prescription totals may vary dramatically from one therapeutic class to another. Instead, it is better to study an analog’s share of its therapeutic class. The goal is to establish what the product’s share might have been if there had been no subsequent, competing market entries or other market events.
Two types of growth curves are often used to model product share curves. One type is the negative exponential curve in which the growth rate declines continuously, and another type is the Gompertz curve (S-shaped curve). For detailed information on the specification and parameter estimation of these curves, please see the book Mathematics of Statistics, (Kenney and Keeping, 1962). The Gompertz curve depicts slow prescription volume growth in the early phase of a product launch followed by accelerated growth as more and more physicians begin to prescribe the product and as word of mouth begins to operate in both the professional and patient communities. After some period of time, the market begins to saturate and growth levels off. The negative exponential curve, on the other hand, illustrates more rapid growth in the early phase. In IMS Health’s experience, the early slow-growth portion of a new pharmaceutical product launch curve is so brief that it usually can be ignored. What is left is a share curve that grows very rapidly at first, and then flattens, i.e., the growth rate constantly decreases over time. By analyzing hundreds of new product growth curves, IMS has found that, typically, a new pharmaceutical product share growth follows a negative exponential curve. By adjusting the negative exponential curve’s parameters, it is possible to generate a curve with the desired shape and asymptotic market share (Figure 2).
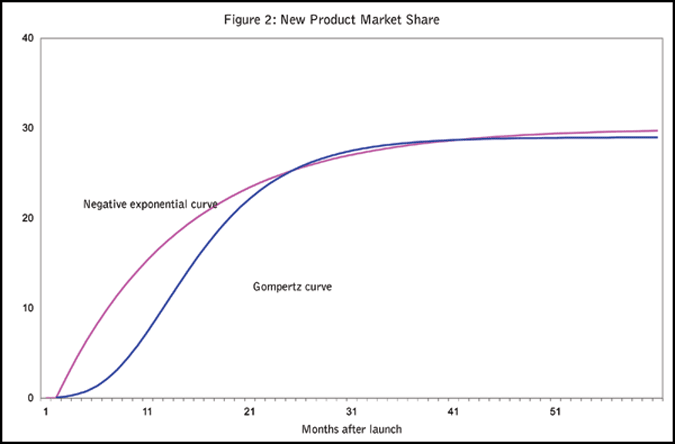
It is not surprising that new pharmaceutical product launch curves have a negative-exponential curve shape with the starting point zero. Early in the product life cycle, there are relatively more new prescribers and patients than in later periods. In addition, promotion spending is generally higher in the first few years. Usually, new product market share grows very slowly after about two to three years post-launch.
At this point, one must also consider how the new product launch will impact the market growth rate by looking into the impact that the analogs had on the expansion of their respective markets.
Market events and impact assessment
Many factors can influence the performance of the market and an individual new product. Some of these factors affect the entire industry (such as the state of the economy, increased unemployment, reduced insurance coverage, demographics and Medicare drug benefits), while other factors impact only specific therapeutic areas (such as practice guidelines and epidemiological trend of the disease, new technologies and new tests, new products in the pipeline, over-the-counter [OTC] switches and patent expirations). Therefore, baseline forecasts for the market volume and for the new product share must be modified in anticipation of the market events and their possible ramifications. For example, the new treatment guidelines that were mentioned earlier for lowering LDL cholesterol should have a positive impact on the growth rate of the cholesterol reducers market.
Let’s again take the cholesterol reducers market as an example. Zocor and Pravachol will lose their patent protections in 2006. In the past, when generics entered the market, they took share only from the branded product within the same molecule. They did not alter the market share trend of the molecule, nor did they expand the treatment market. However, with the pressure to contain drug costs and the practice of managed care plans to require a higher copay for branded products once a generic is available, generics have recently taken share not only from the branded product of the same molecule, but also from products derived from other molecules in the same treatment market. When Mevacor lost its patent in December of 2000, the generic forms not only wiped out Mevacor prescriptions but also took share from other products in the same market and expanded the market share for the total lovastatin molecule (Figure 3).
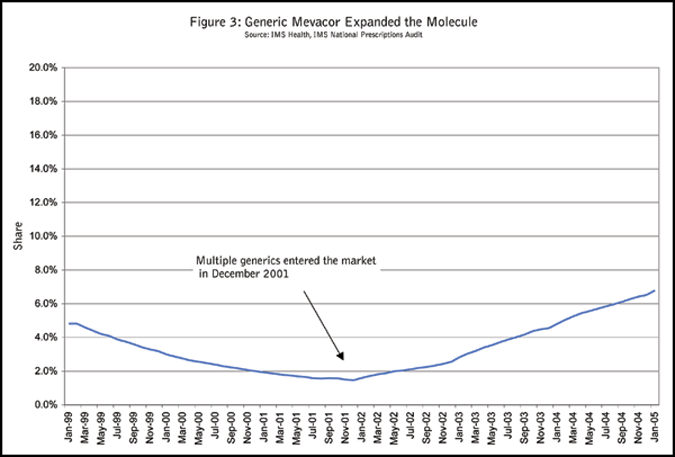
Modifying market forecasts and new product share forecasts based on events
To apply the impact of specific events to the baseline forecast of a therapeutic and to a new product baseline market share, one must quantify the impact of each event, considering:
- timing - that is, when the event is likely to happen;
- magnitude - what is the magnitude of the peak impact of the event;
- time to peak impact - in other words, how long it takes to reach the peak impact;
- probability - or the likelihood that this event will occur.
Because the same event may have a different impact on the market than on the new product, the magnitude and time to peak impact may be different for each. Once the baseline forecasts are thus modified, they are considered “evented” forecasts.
Consider, for example, the impact of statins being switched to OTC status. Since the 10mg strength of Zocor assumed OTC status in the United Kingdom in July 2004, there’s been talk of OTC statins becoming available in the United States. Should that happen, it would have a negative impact on prescription drug volumes as statins go off- patent - and that impact must be calculated as part of a market forecast.
It is most likely that only the lowest strengths (10mg) of statins would go OTC in the United States . Zocor and Pravachol will go off-patent in 2006, and, according to information from IMS Health, their 10mg strengths account for 14 percent and 6 percent of their sales, respectively. Therefore, the total amount at risk is about $0.74B and the estimated impact on the market is about -0.3 percent. The timing of a Zocor and Pravachol OTC switch is likely to be at the end of 2006 when they lose patent protection. The estimated peak impact will be about –0.3 percent on the cholesterol reducers market as a whole and will have no impact on the estimated share of any new product that enters the market subsequently. Based on the recent examples of Claritin OTC and Prilosec OTC, the time to peak impact will be about two to three years. The probability that this event would actually occur is low (10 percent), since an FDA advisory committee recommended against approving Mevacor for OTC status in 2004.
To estimate the prescription volume of a new product, one must multiply the “evented” forecast of the market volume and the “evented” forecast of the new product market share. For example, for a hypothetical statin product to be launched in 2007, based on an analog analysis, the product would likely achieve about 3-5 percent market share and about 8.6 to 14.4 million total prescriptions in 2009.
Promotional resources planning for new product launch
One goal of conducing a forecast of market potential is to be able to determine the optimum level of promotion that should be allocated to launch a new product. To provide evidence-based guidance on promotional resource planning, IMS Health analyzed the professional promotion spending patterns in the first five years after launch for 10 recently- launched products in the United States:
| Product | Therapeutic Area |
| Amerge | Anti-migraine |
| Maxalt | Anti-migraine |
| Meridia | Anti-obesity |
| Xenical | Anti-obesity |
| Comtan | Anti-Parkinson’s |
| Micardis | Anti-hypertension |
| Atacand | Anti-hypertension |
| Actos | Anti-diabetics |
| Avandia | Anti-diabetics |
| Prandin | Anti-diabetics |
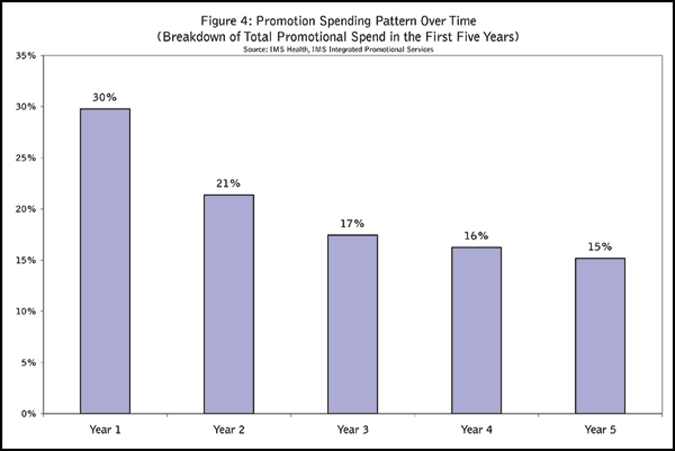
The promotional costs included in the analysis relate to physician detailing, sampling and professional journal advertisement. In the first five years following a launch, the total professional promotion spend is about 15 to 25 percent of the total sales achieved in the same time period. For example, if a product achieved total $2B sales in the first five years, the company spent $300M to $500M in total on promotions in that period. The spending pattern is front-loaded with about 30 percent of the total spent in the first year (Figure 4).
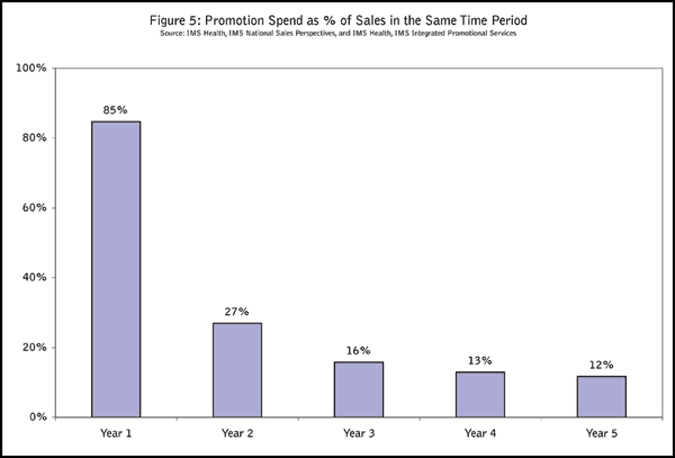
Promotional spend in the first year is about 85 percent of the first year’s sales. Over time, promotional spend drops to about 10 percent of the sales for a given year (Figure 5).
Primary research
Although analogs play a very important role in forecasting new product market potential, primary research with physicians, payers and patients can provide valuable information that may not be available otherwise. And, primary research is even more important than analogs when only sparse secondary data is available or when significant new events are anticipated. Whenever possible, qualitative and/or quantitative primary research should be undertaken with stakeholders. Primary research with doctors can collect data on doctors’ perspectives on product performance and on their satisfaction with existing treatment options, the role of product attributes in their therapy selections, their reactions to the new product based on the product profiles, and their likely usage of the new product.
Similarly, primary research with payers and patients can provide information on price sensitivity and other insights. However, preference share estimated from primary research cannot be treated as a market share estimate for a new product because survey biases do exist. For instance, physicians often overestimate their usage of new products and underestimate their usage of old products. In addition, market share will be influenced by many factors other than physicians’ preference share. These factors include order of entry, promotional support, product uptake rate, competitive promotions and performance, and competitive product launches.
Forecast boundaries
Because of many uncertainties on the future developments of the therapeutic area and environment factors, it is wise to develop pessimistic and optimistic cases in addition to the “most likely” case, in this way providing a forecast boundary. These forecast bounds can be developed based on analog analysis, and/or by changing some of the forecast assumptions such as certain product attributes, or the level of promotional support. Analyzing the promotional response of analogs using promotion mix models can also provide guidance on setting forecasting range and allocate promotional resources needed to launch the product.
Forecasting new product potential within the pharmaceutical industry is not easy, but neither is it impossible. A wealth of resources exist and the methodologies have been well tested.
References
Spyros Makridakis, Steven Wheelwright, and Rob Hyndman, Forecasting: Methods and Applications, 3rd edition. New York : John Wiley, 1998.
J.F. Kenny and E.S. Keeping, Mathematics of Statistics, 3rd edition. Princeton , N.J. : Van Nostrand, 1962.
IMS Health, IMS National Prescription Audit.
IMS Health, IMS National Sales Perspectives.
IMS Health, IMS Integrated Promotional Services.
